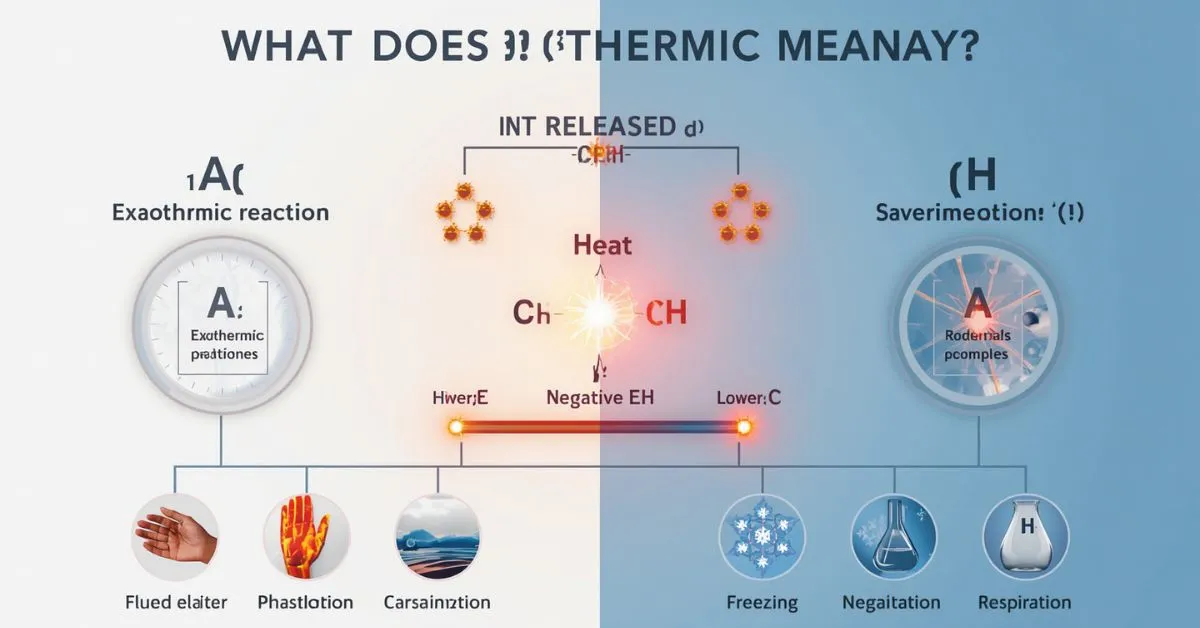
Exothermic means a reaction or process releases energy to its surroundings, usually as heat. In simple terms, heat leaves the system, so the surrounding area often gets warmer. Common exothermic examples include combustion, respiration, hand warmers, freezing, condensation, and many acid-base neutralization reactions.
If you are searching what does exothermic mean, you probably want a fast, clear answer without heavy chemistry jargon. The short answer is simple: an exothermic reaction gives off heat.
But to really understand the term, it helps to know what happens to energy, enthalpy, reactants, products, bonds, the system, and the surroundings. Once those ideas are clear, terms like negative ΔH, activation energy, combustion, and endothermic vs exothermic make much more sense.
This guide explains the meaning in plain English, then builds into examples, comparisons, common mistakes, and practical takeaways.
What does exothermic mean?
In chemistry, exothermic describes a reaction or process that releases energy to the surroundings. Most of the time, that energy is released as heat, although some exothermic reactions also give off light.
Britannica defines exothermic reactions as reactions in which energy as heat is evolved, and Khan Academy explains that exothermic processes release heat and raise the temperature of the immediate surroundings.
Exothermic meaning in simple words
A very simple definition is this:
Exothermic means heat exits the system.
That memory trick helps because the prefix exo- suggests “outside” or “out.” So when a reaction is exothermic, energy goes out from the reacting system into the nearby environment.
System and surroundings
Chemistry often talks about a system and its surroundings. The system is the reaction itself. The surroundings are everything around it, such as the air, water, lab bench, beaker, or your hand touching the container.
In an exothermic change, heat moves from the system to the surroundings, which is why the surroundings often feel warmer.
How exothermic reactions work
To understand exothermic reactions more deeply, you need to look at reactants, products, chemical bonds, enthalpy, and activation energy.
Reactants have more stored energy than products
In an exothermic reaction, the reactants start with more stored chemical energy than the products. As the reaction happens, some of that stored energy is released to the surroundings. That is why the products end up at a lower enthalpy than the reactants.
Monash and LibreTexts both explain that, for exothermic reactions, the combined enthalpy of the products is lower than the combined enthalpy of the reactants.
Bond breaking and bond making
A common source of confusion is chemical bonds. Breaking bonds requires energy, while making bonds releases energy. A reaction is exothermic when the energy released by forming new bonds in the products is greater than the energy needed to break the old bonds in the reactants. That net release of energy is what you feel as heat.
Activation energy still matters
Even exothermic reactions often need a small push to start. That starting push is called activation energy. For example, wood burning in a fire is exothermic, but it still needs a spark or flame to begin. After the reaction starts, more energy is released than was needed to get it going.
What does negative ΔH mean?
In thermochemistry, the energy change of a reaction is often written as ΔH, the enthalpy change. For an exothermic reaction, ΔH is negative. That negative sign shows that heat leaves the system and the enthalpy of the system decreases.
Exothermic vs endothermic
The term most often compared with exothermic is endothermic. These are opposite ideas.
- Exothermic reactions release heat to the surroundings.
- Endothermic reactions absorb heat from the surroundings.
Quick comparison table
| Feature | Exothermic | Endothermic |
|---|---|---|
| Heat flow | Out of the system | Into the system |
| Effect on surroundings | Surroundings get warmer | Surroundings get cooler |
| Enthalpy change | Negative ΔH | Positive ΔH |
| Products vs reactants | Products have lower enthalpy | Products have higher enthalpy |
| Simple memory trick | Exo = exit | Endo = enter |
| Common example | Burning fuel | Melting ice |
This comparison matches standard thermochemistry teaching: exothermic reactions are energetically “downhill,” while endothermic reactions are “uphill.”
Easy memory trick
A simple way to remember the difference is:
- Exo = exit
- Endo = enter
If heat exits the system, the process is exothermic. If heat enters the system, the process is endothermic.
Common exothermic examples
Examples make the meaning much easier to remember. Exothermic changes are not limited to one type of chemistry. They appear in combustion, oxidation, phase changes, dissolving, neutralization, and biological processes.
1) Combustion
Combustion is one of the clearest exothermic reactions. When petrol, wood, coal, natural gas, or a candle burns, energy is released to the environment as heat and often light. Monash specifically uses burning petrol as an example of an exothermic combustion reaction.
2) Respiration
Respiration is the biological process that lets cells oxidize food molecules and release energy. Britannica notes that the chemical energy in food is converted by the body into mechanical energy and heat, which makes respiration an everyday exothermic example.
3) Hand warmers
Many hand warmers work because of an exothermic process or reaction. American Chemical Society materials describe hand warmers as exothermic and explain that they warm up because more energy is released in product formation than is needed to break bonds in the reactants.
Some common disposable hand warmers use oxidation of iron, while others use crystallization systems such as sodium acetate.
4) Freezing and condensation
Exothermic does not always mean flames. Freezing and condensation are also exothermic processes. LibreTexts explains that freezing, condensation, and deposition release heat to the surroundings because they move toward more ordered states. Khan Academy also gives condensation of water vapor into rain as an exothermic process.
5) Acid-base neutralization
Many neutralization reactions are exothermic. When an acid reacts with a base, energy is commonly released to the surroundings. This is why the temperature often rises during strong acid-strong base neutralization in lab experiments and titrations.
6) Dissolving calcium chloride
Not every exothermic change is a chemical reaction in the narrow sense. Some dissolving processes are exothermic too. Calcium chloride is a classic example because it releases heat when it dissolves in water, which is why it is used in some instant hot packs.
How to tell if a reaction is exothermic
If you are solving homework, doing a lab, or looking at a thermochemical equation, here are the best ways to identify an exothermic process.
The surroundings get warmer
The most obvious sign is that the temperature of the surroundings rises. A beaker may feel warm, the air near the reaction may heat up, or a thermometer may show an increase. Khan Academy describes this as a key sign of exothermic processes.
ΔH is negative
If a reaction has ΔH < 0, it is exothermic. This is one of the clearest ways to identify it in chemistry class, especially in energy diagrams, enthalpy calculations, and thermochemical equations.
Products sit lower on an energy profile diagram
On an energy profile diagram, the products of an exothermic reaction are lower than the reactants. The curve still rises first because of activation energy, but the final energy level is lower.
A calorimeter measures released heat
In more advanced chemistry, a calorimeter is used to measure heat transfer. LibreTexts explains that during exothermic combustion, the temperature of the calorimeter and surrounding bath rises, allowing the released heat to be calculated.
Common mistakes people make
Mistake 1: Thinking exothermic always means fire
That is not true. Fire is exothermic, but so are freezing, condensation, and some dissolving processes. Exothermic is about energy leaving the system, not just visible flames.
Mistake 2: Thinking bond breaking releases energy
This is one of the biggest chemistry mistakes. Bond breaking requires energy. Bond formation releases energy. Exothermic reactions happen when bond making releases more than bond breaking uses.
Mistake 3: Mixing up system and surroundings
Students often look only at the reaction itself and forget the surroundings. The key question is not “Is the reaction hot?” but “Where is the heat going?” If heat goes from the system to the surroundings, it is exothermic.
Mistake 4: Confusing exothermic with exoergic
In basic chemistry lessons, “exothermic” is usually enough. But technically, exoergic is broader and can refer to energy released in forms other than heat. Britannica notes this distinction. For most school-level explanations, though, exothermic is the term you need.
Why exothermic matters in real life
Exothermic reactions matter far beyond the classroom. They shape how we cook, travel, stay warm, power machines, and understand weather, biology, and industrial chemistry.
In daily life, exothermic processes appear in candles, stoves, engines, fires, respiration, and hot packs. In labs, they appear in neutralization, calorimetry, and enthalpy experiments.
In industry, they matter in combustion, fuel energy, temperature control, and reaction safety. In physical chemistry, they connect to thermochemical equations, heat of combustion, enthalpy of solution, Hess’s law, and energy diagrams.
Practical takeaways
If you want one strong takeaway, remember this:
Exothermic means energy leaves the system and goes into the surroundings.
That one sentence helps you answer most beginner questions. Then add three more memory anchors:
- Heat exits the system
- ΔH is negative
- The surroundings usually get warmer
FAQs
Is exothermic hot or cold?
Exothermic processes usually make the surroundings feel warmer because heat is released from the system to the surroundings.
What is the simplest definition of exothermic?
The simplest definition is: an exothermic reaction releases heat.
Is freezing exothermic?
Yes. Freezing is exothermic because liquid turning into solid releases heat to the surroundings.
Is condensation exothermic?
Yes. Condensation is exothermic because gas changing into liquid releases heat.
Why is ΔH negative in an exothermic reaction?
Because the system loses heat to the surroundings, the system’s enthalpy decreases, so ΔH is negative.
What is the difference between exothermic and endothermic?
Exothermic reactions release heat, while endothermic reactions absorb heat.
Is burning wood exothermic?
Yes. Burning wood is a combustion reaction, and combustion releases energy as heat and light.
Are hand warmers exothermic?
Yes. Many hand warmers work through an exothermic process or reaction that releases heat.
Conclusion
So, what does exothermic mean? It means a reaction or process releases energy to its surroundings, usually in the form of heat. That is why exothermic changes often make nearby objects feel warmer.
Once you connect the term to system vs surroundings, negative ΔH, bond making and bond breaking, and easy examples like combustion, freezing, condensation, respiration, neutralization, and hand warmers, the concept becomes much easier to remember.
Click Here To Read About: What Does a Poison Tree Tattoo Mean?

Hi, I’m Clara Lexis from Meanvia.com. I break down words and expressions so they’re easy to understand and enjoyable to learn. My mission is simple: make language approachable and fun, one word at a time.